Label materials for Pharmacy
Labeling Solutions for Pharmacy Product
Pharmacy labeling plays a critical role in ensuring patient safety, regulatory compliance, and product integrity across prescription drugs, over-the-counter medications, and clinical packaging. Labels must remain securely affixed and legible throughout the product’s lifecycle—from manufacturing and distribution to dispensing and patient use—often under demanding conditions such as refrigeration, sterilization, and repeated handling.
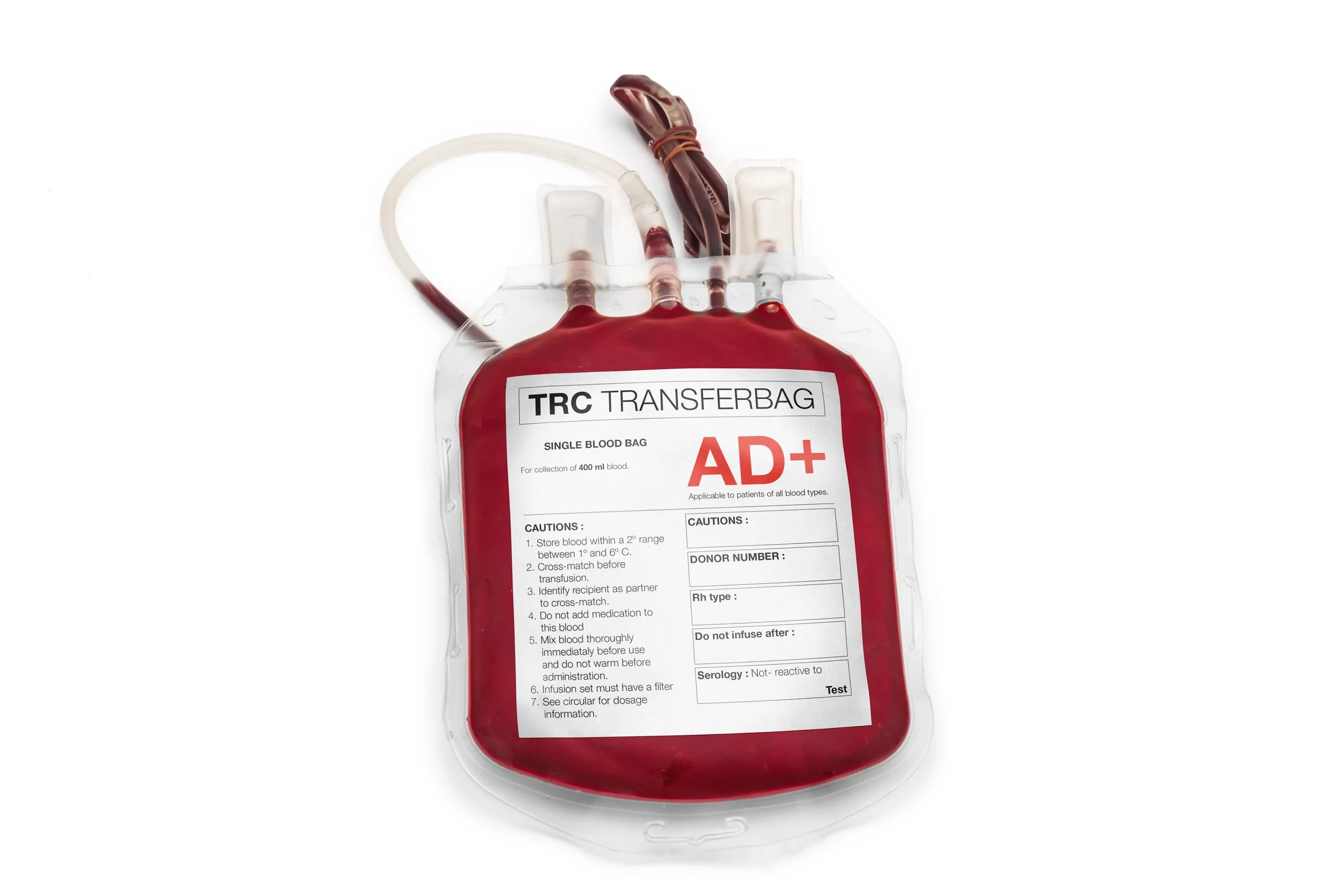
Pharmaceutical labels are applied to a wide range of substrates including glass vials, plastic syringes, blister packs, infusion bags, and carton boxes. Each format presents unique challenges: small diameters, curved surfaces, low surface energy plastics, and exposure to chemicals or moisture. Label materials must be engineered for low migration, chemical inertness, and sterility compatibility, ensuring they do not compromise the safety or efficacy of the drug product.
In regulated environments, pharmacy labels must meet stringent standards such as FDA, EU MDR, and USP guidelines, often requiring tamper-evident features, anti-counterfeit elements, and presence verification technologies. These safeguards help prevent medication errors, support traceability, and protect against diversion or falsification.
Operationally, pharmacy labels must support thermal transfer printing, laser coding, and variable data integration for batch numbers, expiration dates, and barcodes. In hospital and clinical settings, labels may also include color-coded warnings, multilingual instructions, and patient-specific data. For blood bags, biologics, and temperature-sensitive drugs, label adhesives must perform reliably in cold storage and withstand sterilization processes without delamination or ink degradation.
As pharmaceutical packaging evolves, so do labeling demands. Today’s pharmacy labels incorporate smart technologies, sustainable materials, and modular formats that adapt to diverse dosage forms and global compliance requirements. Whether applied to a single-dose vial or a multi-dose blister pack, pharmacy labels are a vital interface between product, provider, and patient—delivering clarity, safety, and trust in every application.
Product Code: QB4389
Adhesive Code: QJ285
Facestock: Paper (Semi Gloss)
Basis Weight of Facestock (g/m²): 80
Facestock Color: White
Adhesive Type: Acrylic
Adhesive Application: FDA 175.105
Liner: White Glassine Paper
Liner Basis Weight (g/m²): 60
Sample Ready: Yes
Batch No: 1
Product Code: QB6389
Adhesive Code: IJ285
Facestock: Paper (Semi Gloss)
Basis Weight of Facestock (g/m²): 80
Facestock Color: White
Adhesive Type: Rubber based
Adhesive Application:
1. FDA 175.106
2. The migration requirement of EU Framework Regulation—(EC) No 1935/2004
Liner: White Glassine Paper
Liner Basis Weight (g/m²): 60
Sample Ready: Yes
Batch No: 1
Product Code: QB2341
Adhesive Code: QJ285
Facestock: Paper (Light Weight Semi Gloss)
Basis Weight of Facestock (g/m²): 60
Facestock Color: White
Adhesive Type: Acrylic
Adhesive Application: FDA 175.105
Liner: White Glassine Paper
Liner Basis Weight (g/m²): 60
Sample Ready: Yes
Batch No: 1
Product Code: QB6341P
Adhesive Code: IJ285
Facestock: Paper (Light Weight Semi Gloss)
Basis Weight of Facestock (g/m²): 64
Facestock Color: White (FSC approval)
Adhesive Type: Rubber based
Adhesive Application: FDA 175.105
Liner: White Glassine Paper
Liner Basis Weight (g/m²): 60
Sample Ready: Yes
Batch No: 1